What is Global Medical Device Testing and Certification Market?
The Global Medical Device Testing and Certification Market is a crucial component of the healthcare industry, ensuring that medical devices meet the necessary safety and quality standards before they reach consumers. This market involves a series of rigorous processes that include testing, inspection, and certification of medical devices. These processes are essential to verify that devices comply with international and national regulations, which are designed to protect patient safety and ensure product efficacy. The market is driven by the increasing complexity of medical devices, advancements in technology, and stringent regulatory requirements across different regions. As medical devices become more sophisticated, the need for comprehensive testing and certification becomes even more critical. This market not only helps manufacturers meet regulatory requirements but also enhances their credibility and marketability by ensuring that their products are safe and reliable. The global nature of this market means that it involves a wide range of stakeholders, including manufacturers, regulatory bodies, testing laboratories, and certification organizations, all working together to ensure that medical devices are safe for use. The market is expected to grow as the demand for medical devices continues to rise, driven by factors such as an aging population, increasing prevalence of chronic diseases, and technological advancements in the healthcare sector.

Testing, Inspection, Certification in the Global Medical Device Testing and Certification Market:
Testing, inspection, and certification are the three pillars of the Global Medical Device Testing and Certification Market. Testing involves a series of procedures to evaluate the performance, safety, and efficacy of medical devices. This can include mechanical testing, electrical safety testing, biocompatibility testing, and software validation, among others. Each type of test is designed to assess different aspects of a device's functionality and safety. For instance, mechanical testing evaluates the physical properties of a device, such as its strength and durability, while electrical safety testing ensures that the device operates safely under various electrical conditions. Biocompatibility testing is crucial for devices that come into contact with the human body, as it assesses the potential for adverse biological reactions. Software validation is increasingly important as more devices incorporate complex software systems. Inspection, on the other hand, involves a thorough examination of the manufacturing process and the final product to ensure compliance with regulatory standards. This can include on-site inspections of manufacturing facilities, as well as detailed examinations of the devices themselves. Inspectors look for any deviations from the approved design and manufacturing processes, as well as any potential safety issues. Certification is the final step in the process, where a third-party organization verifies that a device meets all necessary regulatory requirements and standards. This certification is often required for a device to be marketed and sold in different regions. The certification process can vary depending on the type of device and the regulatory requirements of the target market. In many cases, certification involves a combination of testing and inspection results, along with a review of the manufacturer's quality management system. The certification process provides assurance to both manufacturers and consumers that a device is safe and effective for its intended use. Overall, the testing, inspection, and certification processes are essential for ensuring the safety and quality of medical devices, and they play a critical role in the global healthcare industry.
Large Enterprises, SMEs in the Global Medical Device Testing and Certification Market:
The Global Medical Device Testing and Certification Market plays a significant role in both large enterprises and small and medium-sized enterprises (SMEs). For large enterprises, which often have extensive product lines and operate in multiple international markets, the testing and certification process is a critical component of their operations. These companies typically have the resources to invest in comprehensive testing and certification processes, which can help them maintain compliance with various international standards and regulations. By ensuring that their products meet the necessary safety and quality standards, large enterprises can enhance their reputation and credibility in the market, which can lead to increased sales and market share. Additionally, the certification process can help large enterprises identify potential issues with their products before they reach the market, reducing the risk of costly recalls and damage to their brand reputation. For SMEs, the testing and certification process can be more challenging due to limited resources and expertise. However, it is equally important for these companies to ensure that their products meet the necessary safety and quality standards. SMEs can benefit from the testing and certification process by gaining access to new markets and increasing their competitiveness. By obtaining certification for their products, SMEs can demonstrate their commitment to quality and safety, which can help them build trust with customers and differentiate themselves from competitors. Additionally, the certification process can provide SMEs with valuable insights into their products and manufacturing processes, helping them identify areas for improvement and innovation. Overall, the Global Medical Device Testing and Certification Market provides essential support to both large enterprises and SMEs, helping them ensure the safety and quality of their products and succeed in the competitive global healthcare market.
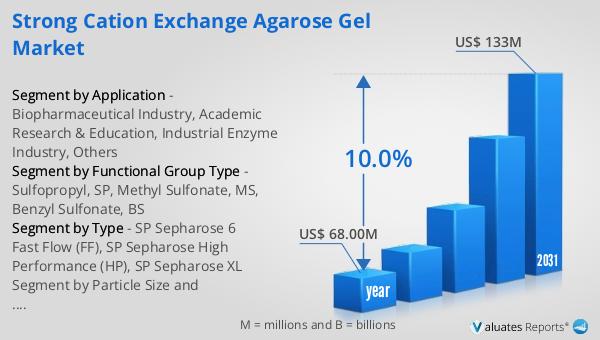
Global Medical Device Testing and Certification Market Outlook:
Our research indicates that the global market for medical devices is projected to reach approximately $603 billion in 2023, with an anticipated growth rate of 5% annually over the next six years. This growth is driven by several factors, including technological advancements, an aging population, and the increasing prevalence of chronic diseases. As the demand for medical devices continues to rise, the importance of testing and certification becomes even more critical. Ensuring that medical devices meet the necessary safety and quality standards is essential for protecting patient safety and ensuring product efficacy. The testing and certification process helps manufacturers meet regulatory requirements and enhances their credibility and marketability. By ensuring that their products are safe and reliable, manufacturers can build trust with consumers and gain a competitive edge in the market. The global nature of the medical device market means that manufacturers must navigate a complex landscape of international and national regulations, making the testing and certification process even more important. As the market continues to grow, the demand for testing and certification services is expected to increase, providing opportunities for companies in this sector to expand their offerings and reach new customers. Overall, the Global Medical Device Testing and Certification Market is a vital component of the healthcare industry, ensuring that medical devices are safe and effective for use.
| Report Metric | Details |
| Report Name | Medical Device Testing and Certification Market |
| Accounted market size in year | US$ 603 billion |
| CAGR | 5% |
| Base Year | year |
| Segment by Type |
|
| Segment by Application |
|
| By Region |
|
| By Company | BSI Group, Eurofins Scientific, TUV Rheinland, UL LLC, Dekra Testing and Certification, Institute for testing and Certification, Intertek Group, SGS SA, Bureau Veritas, Element Materials Technology, Avomeen, Gateway Analytical, WuXi AppTec, Cigniti, Charles River, MET Labs, Noble Life Sciences |
| Forecast units | USD million in value |
| Report coverage | Revenue and volume forecast, company share, competitive landscape, growth factors and trends |