What is Global Pharmaceutical Grade Nervous Acid Products Market?
The Global Pharmaceutical Grade Nervous Acid Products Market is a specialized segment within the broader pharmaceutical industry, focusing on the production and distribution of high-purity nervous acid. Nervous acid, a long-chain fatty acid, is primarily derived from natural sources and is known for its potential therapeutic benefits, particularly in neurological health. The market for pharmaceutical-grade nervous acid products is driven by the increasing demand for advanced therapeutic solutions targeting neurological disorders, such as Alzheimer's disease, multiple sclerosis, and other neurodegenerative conditions. As research continues to uncover the potential benefits of nervous acid in promoting brain health and cognitive function, pharmaceutical companies are investing in the development of innovative products that incorporate this compound. The market is characterized by a growing emphasis on quality and purity, with manufacturers striving to meet stringent regulatory standards to ensure the safety and efficacy of their products. Additionally, the rising prevalence of neurological disorders globally, coupled with an aging population, is expected to fuel the demand for pharmaceutical-grade nervous acid products, making this market a promising area for growth and innovation in the coming years.
Content >95%, Content >90% in the Global Pharmaceutical Grade Nervous Acid Products Market:
In the Global Pharmaceutical Grade Nervous Acid Products Market, the purity of the product is a critical factor that influences its application and efficacy. Products with a content greater than 95% are considered to be of the highest quality, offering superior therapeutic benefits due to their high concentration of active ingredients. These products are primarily used in the development of advanced pharmaceuticals aimed at treating complex neurological disorders. The high purity ensures that the nervous acid can effectively interact with biological systems, enhancing its potential to improve cognitive function and support neurological health. On the other hand, products with a content greater than 90% are also significant in the market, offering a balance between efficacy and cost. While slightly less concentrated than their >95% counterparts, these products still provide substantial therapeutic benefits and are often used in the formulation of health supplements and over-the-counter products. The choice between >95% and >90% content products often depends on the specific application and the desired outcome. For instance, pharmaceutical companies developing prescription medications for severe neurological conditions may opt for the >95% products to ensure maximum efficacy, while manufacturers of dietary supplements may choose the >90% products to offer consumers an effective yet affordable option. The market dynamics for these products are influenced by factors such as regulatory requirements, production costs, and consumer demand. As the understanding of nervous acid's role in neurological health continues to evolve, the demand for both >95% and >90% content products is expected to grow, driven by the need for effective and accessible therapeutic solutions. Manufacturers are likely to focus on optimizing production processes to enhance the purity and quality of their products while maintaining cost-effectiveness. This balance between quality and affordability is crucial in ensuring that pharmaceutical-grade nervous acid products can reach a broad audience, including both healthcare providers and consumers seeking to improve their neurological health. The ongoing research and development efforts in this field are expected to lead to the introduction of new and innovative products, further expanding the market and providing more options for those in need of effective neurological therapies.
Drugs, Health Products, Other in the Global Pharmaceutical Grade Nervous Acid Products Market:
The Global Pharmaceutical Grade Nervous Acid Products Market finds its application in various areas, including drugs, health products, and other sectors. In the pharmaceutical industry, nervous acid is primarily used in the development of drugs aimed at treating neurological disorders. Its potential to enhance cognitive function and support brain health makes it a valuable component in medications targeting conditions such as Alzheimer's disease, Parkinson's disease, and multiple sclerosis. Pharmaceutical companies are investing in research to explore the full therapeutic potential of nervous acid, leading to the development of innovative drugs that can improve the quality of life for patients suffering from these debilitating conditions. In the health products sector, nervous acid is often incorporated into dietary supplements and functional foods designed to support overall brain health and cognitive function. These products are popular among consumers seeking to maintain mental acuity and prevent age-related cognitive decline. The demand for such health products is driven by an increasing awareness of the importance of brain health and the desire to take proactive measures to preserve cognitive function. Additionally, nervous acid is used in other sectors, such as cosmetics and personal care products, where its anti-inflammatory and antioxidant properties are valued for promoting skin health and reducing the signs of aging. The versatility of nervous acid in various applications highlights its potential as a multifunctional ingredient with broad market appeal. As research continues to uncover new benefits and applications for nervous acid, its usage is expected to expand across different industries, further driving the growth of the Global Pharmaceutical Grade Nervous Acid Products Market. Manufacturers are likely to focus on developing innovative formulations that leverage the unique properties of nervous acid to meet the diverse needs of consumers and healthcare providers. This ongoing innovation and diversification of applications will play a crucial role in the market's expansion and success in the coming years.
Global Pharmaceutical Grade Nervous Acid Products Market Outlook:
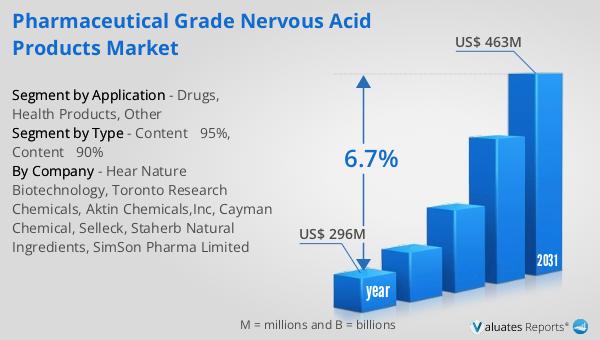
The global market for Pharmaceutical Grade Nervous Acid Products was valued at approximately $296 million in 2024 and is anticipated to grow to a revised size of $463 million by 2031, reflecting a compound annual growth rate (CAGR) of 6.7% during the forecast period. This growth is indicative of the increasing demand for high-quality nervous acid products, driven by their potential therapeutic benefits in treating neurological disorders. In comparison, the broader global pharmaceutical market was valued at $1,475 billion in 2022, with an expected CAGR of 5% over the next six years. This comparison highlights the relatively faster growth rate of the nervous acid products market, underscoring its potential as a niche yet rapidly expanding segment within the pharmaceutical industry. Meanwhile, the chemical drug market, a significant component of the pharmaceutical industry, was estimated to grow from $1,005 billion in 2018 to $1,094 billion in 2022. This steady growth reflects the ongoing demand for chemical-based therapeutic solutions, although the growth rate is slightly lower than that of the nervous acid products market. The data suggests that while the overall pharmaceutical market continues to expand, there is a particular interest in specialized segments like nervous acid products, which offer unique therapeutic benefits and opportunities for innovation. As the market for pharmaceutical-grade nervous acid products continues to grow, it is expected to attract increased investment and research efforts, further driving its development and integration into mainstream therapeutic solutions.
| Report Metric | Details |
| Report Name | Pharmaceutical Grade Nervous Acid Products Market |
| Accounted market size in year | US$ 296 million |
| Forecasted market size in 2031 | US$ 463 million |
| CAGR | 6.7% |
| Base Year | year |
| Forecasted years | 2025 - 2031 |
| Segment by Type |
|
| Segment by Application |
|
| Consumption by Region |
|
| By Company | Hear Nature Biotechnology, Toronto Research Chemicals, Aktin Chemicals,Inc, Cayman Chemical, Selleck, Staherb Natural Ingredients, SimSon Pharma Limited |
| Forecast units | USD million in value |
| Report coverage | Revenue and volume forecast, company share, competitive landscape, growth factors and trends |