What is Global Thrombopoietin Receptor Market?
The Global Thrombopoietin Receptor Market is a specialized segment within the broader pharmaceutical and biotechnology industries, focusing on the development and commercialization of drugs that target thrombopoietin receptors. These receptors play a crucial role in the regulation of platelet production in the body, which is essential for blood clotting and wound healing. The market is driven by the increasing prevalence of conditions such as chronic immune thrombocytopenia (ITP), aplastic anemia, and other disorders that lead to low platelet counts. As awareness and diagnosis of these conditions improve, the demand for effective treatments is rising. The market is characterized by ongoing research and development efforts aimed at creating more effective and safer drugs. Companies operating in this space are investing heavily in clinical trials and regulatory approvals to bring new products to market. The competitive landscape is marked by both established pharmaceutical giants and emerging biotech firms, all vying for a share of this growing market. As the global population ages and the incidence of related health conditions increases, the Global Thrombopoietin Receptor Market is expected to continue its growth trajectory, offering new opportunities for innovation and patient care.
Eltrombopag Olamine, GSK-2285921, Romiplostim, STST-4, Others in the Global Thrombopoietin Receptor Market:
Eltrombopag Olamine, GSK-2285921, Romiplostim, STST-4, and other drugs are key players in the Global Thrombopoietin Receptor Market, each contributing uniquely to the treatment landscape for thrombocytopenia and related conditions. Eltrombopag Olamine, marketed under the brand name Promacta, is an oral thrombopoietin receptor agonist that stimulates platelet production. It is widely used for patients with chronic immune thrombocytopenia (ITP) who have not responded adequately to other treatments. Its oral administration offers a convenient option for patients, enhancing compliance and quality of life. GSK-2285921 is another promising candidate in this market, developed by GlaxoSmithKline. Although still in the experimental stages, it represents the ongoing innovation within the industry, aiming to provide more effective solutions with fewer side effects. Romiplostim, marketed as Nplate, is an injectable thrombopoietin receptor agonist that mimics the action of natural thrombopoietin. It is particularly effective for patients with chronic ITP and is administered via subcutaneous injection, offering an alternative for those who may not respond to oral medications. STST-4 is a newer entrant in the market, reflecting the continuous research and development efforts to expand treatment options. While specific details about STST-4 are limited, its inclusion in the market highlights the dynamic nature of drug development in this field. Other drugs in the market also contribute to the diverse array of treatment options available to patients, each with unique mechanisms of action and administration routes. The competition among these drugs drives innovation, leading to improved patient outcomes and expanding the therapeutic arsenal available to healthcare providers. As research progresses, the Global Thrombopoietin Receptor Market is poised to offer even more advanced treatments, addressing unmet medical needs and enhancing the quality of life for patients worldwide.
Clinic, Hospital, Others in the Global Thrombopoietin Receptor Market:
The Global Thrombopoietin Receptor Market finds significant application in various healthcare settings, including clinics, hospitals, and other medical facilities. In clinics, these drugs are often used for outpatient management of conditions like chronic immune thrombocytopenia (ITP) and aplastic anemia. Clinics provide a more accessible and less intimidating environment for patients, making them an ideal setting for administering treatments like Eltrombopag Olamine, which can be taken orally. This convenience allows patients to maintain their daily routines with minimal disruption, improving adherence to treatment regimens. Hospitals, on the other hand, play a crucial role in the management of more severe cases or complications arising from low platelet counts. Inpatient care may be necessary for patients requiring close monitoring or those experiencing significant bleeding risks. Drugs like Romiplostim, which require subcutaneous injections, are often administered in hospital settings where healthcare professionals can closely monitor patient responses and adjust dosages as needed. Hospitals also serve as centers for clinical trials and research, contributing to the development of new therapies and expanding the knowledge base surrounding thrombopoietin receptor agonists. Beyond clinics and hospitals, other healthcare facilities, such as specialized treatment centers and research institutions, also utilize these drugs. These settings often focus on specific patient populations or conduct advanced research to explore new therapeutic avenues. The versatility of thrombopoietin receptor agonists allows them to be integrated into various treatment protocols, catering to the diverse needs of patients across different healthcare environments. As the Global Thrombopoietin Receptor Market continues to evolve, its applications in these areas are expected to expand, driven by ongoing research, technological advancements, and a growing understanding of platelet-related disorders. This expansion will likely lead to improved patient outcomes, more personalized treatment approaches, and a broader impact on public health.
Global Thrombopoietin Receptor Market Outlook:
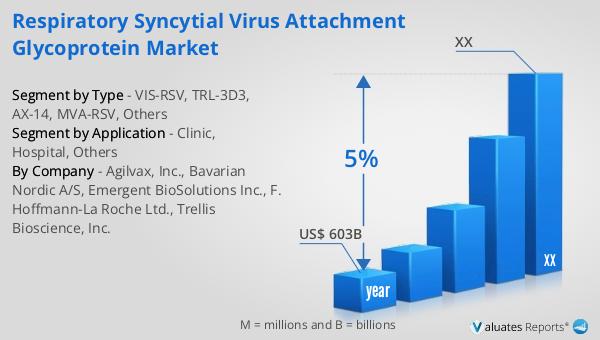
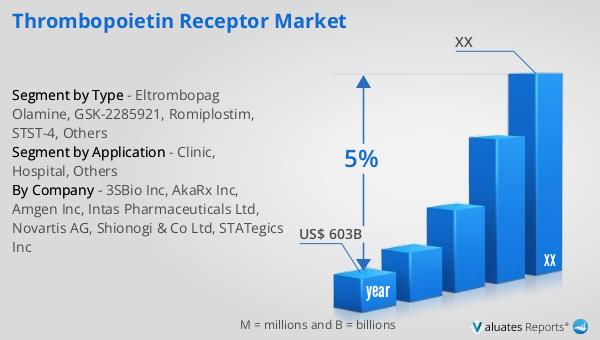
Our research indicates that the global market for medical devices, which includes the Global Thrombopoietin Receptor Market, is projected to reach approximately $603 billion in 2023. This substantial market size reflects the growing demand for innovative medical solutions and the increasing prevalence of chronic health conditions worldwide. Over the next six years, the market is expected to grow at a compound annual growth rate (CAGR) of 5%. This steady growth trajectory underscores the importance of continuous innovation and development within the medical device sector. The expansion of this market is driven by several factors, including advancements in technology, increased healthcare spending, and a rising focus on improving patient outcomes. As the population ages and the incidence of chronic diseases rises, the demand for effective medical devices and treatments is set to increase. This growth presents significant opportunities for companies operating in the Global Thrombopoietin Receptor Market to expand their product offerings and reach new patient populations. The competitive landscape will likely intensify as more players enter the market, driving further innovation and potentially leading to more cost-effective and accessible treatment options for patients. Overall, the outlook for the Global Thrombopoietin Receptor Market within the broader medical device industry is positive, with promising prospects for continued growth and development.
| Report Metric | Details |
| Report Name | Thrombopoietin Receptor Market |
| Accounted market size in year | US$ 603 billion |
| CAGR | 5% |
| Base Year | year |
| Segment by Type |
|
| Segment by Application |
|
| By Region |
|
| By Company | 3SBio Inc, AkaRx Inc, Amgen Inc, Intas Pharmaceuticals Ltd, Novartis AG, Shionogi & Co Ltd, STATegics Inc |
| Forecast units | USD million in value |
| Report coverage | Revenue and volume forecast, company share, competitive landscape, growth factors and trends |