What is Global Professional Skin Care Product Market?
The Global Professional Skin Care Product Market is a dynamic and expansive sector that caters to the diverse needs of consumers seeking advanced skincare solutions. This market encompasses a wide range of products specifically designed for professional use, including those used in spas, dermatology clinics, and beauty salons. These products are formulated to address various skin concerns, such as aging, acne, pigmentation, and sensitivity, offering targeted treatments that go beyond everyday skincare routines. The market is driven by increasing consumer awareness about skincare, the rising demand for anti-aging products, and the growing popularity of personalized skincare solutions. Additionally, technological advancements in skincare formulations and the incorporation of natural and organic ingredients have further fueled market growth. Key players in this market are continuously innovating to meet the evolving preferences of consumers, ensuring that professional skincare products remain at the forefront of the beauty industry. As a result, the Global Professional Skin Care Product Market is poised for sustained growth, offering a plethora of opportunities for both established brands and emerging players.
Moisturizers, Cream, Lotion, Powders, Sprays, Masks, Others in the Global Professional Skin Care Product Market:
Moisturizers, creams, lotions, powders, sprays, masks, and other products form the backbone of the Global Professional Skin Care Product Market, each serving unique purposes and catering to specific skin needs. Moisturizers are essential for maintaining skin hydration and are available in various formulations to suit different skin types. They help in retaining moisture, preventing dryness, and providing a protective barrier against environmental aggressors. Creams, often richer in texture, are designed to deliver intensive nourishment and are particularly beneficial for dry and mature skin. They are infused with active ingredients that target specific concerns such as wrinkles, fine lines, and loss of elasticity. Lotions, being lighter than creams, are ideal for everyday use and are quickly absorbed by the skin, making them suitable for normal to oily skin types. Powders, although less common in professional skincare, are used for their mattifying properties and are often incorporated into products aimed at controlling excess oil and shine. Sprays, on the other hand, offer a convenient way to refresh and hydrate the skin throughout the day, often containing soothing and calming ingredients like aloe vera and chamomile. Masks are a staple in professional skincare, providing intensive treatment for various skin issues. They come in different forms, such as clay, sheet, and peel-off masks, each offering unique benefits like deep cleansing, hydration, and brightening. Other products in this market include serums, exfoliants, and toners, each playing a crucial role in a comprehensive skincare regimen. Serums are concentrated formulations that deliver potent active ingredients deep into the skin, targeting specific concerns like pigmentation and dullness. Exfoliants help in removing dead skin cells, promoting cell turnover, and revealing a smoother, more radiant complexion. Toners, often used after cleansing, help in balancing the skin's pH levels and preparing it for subsequent treatments. The diversity of products in the Global Professional Skin Care Product Market ensures that there is something for everyone, regardless of their skin type or concern. This variety not only caters to individual preferences but also allows professionals to customize treatments, offering personalized skincare solutions that address the unique needs of each client.
Neutral Skin, Dry Skin, Oily Skin, Mixed Skin, Sensitive Skin in the Global Professional Skin Care Product Market:
The usage of Global Professional Skin Care Products varies significantly across different skin types, including neutral, dry, oily, mixed, and sensitive skin, each requiring tailored approaches to achieve optimal results. For neutral skin, which is generally well-balanced and not prone to extreme conditions, professional skincare products focus on maintaining this balance and preventing future issues. Products such as lightweight moisturizers, gentle cleansers, and broad-spectrum sunscreens are commonly used to preserve the skin's natural state. Dry skin, characterized by a lack of moisture and a tendency to flake, benefits from rich, hydrating products like creams and oils that provide deep nourishment and restore the skin's barrier function. Ingredients such as hyaluronic acid, glycerin, and ceramides are often included in these formulations to attract and retain moisture. Oily skin, which produces excess sebum, requires products that control oil production without stripping the skin of its natural moisture. Lightweight, non-comedogenic moisturizers, mattifying powders, and clay masks are popular choices for managing shine and preventing breakouts. Mixed skin, which exhibits characteristics of both dry and oily skin, poses a unique challenge, requiring a combination of products that address the varying needs of different areas of the face. For instance, a balancing toner might be used to control oil in the T-zone, while a hydrating serum is applied to drier areas. Sensitive skin, prone to irritation and redness, demands gentle, soothing products that minimize the risk of adverse reactions. Formulations free from fragrances, alcohol, and harsh chemicals are preferred, with ingredients like chamomile, aloe vera, and calendula providing calming effects. The Global Professional Skin Care Product Market offers a wide array of options for each skin type, enabling consumers and professionals alike to tailor their skincare routines to achieve healthy, radiant skin.
Global Professional Skin Care Product Market Outlook:
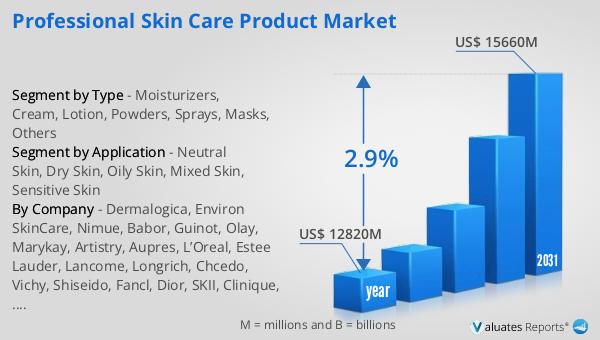
The global market for Professional Skin Care Products was valued at $12,820 million in 2024 and is anticipated to grow to a revised size of $15,660 million by 2031, reflecting a compound annual growth rate (CAGR) of 2.9% over the forecast period. This growth is driven by several factors, including increasing consumer awareness about skincare, the rising demand for anti-aging products, and the growing popularity of personalized skincare solutions. Major players in this market include renowned brands such as L'Oreal, P&G, Estee Lauder, Shiseido, Unilever, LVMH, Chanel, Amore Pacific Group, LG Group, and Kanabo. These companies collectively hold about 38% of the market share, underscoring their significant influence and leadership in the industry. Their commitment to innovation and quality has enabled them to maintain a competitive edge, offering a diverse range of products that cater to the evolving needs of consumers. As the market continues to expand, these key manufacturers are poised to capitalize on emerging trends and opportunities, further solidifying their positions in the global professional skincare landscape.
| Report Metric | Details |
| Report Name | Professional Skin Care Product Market |
| Accounted market size in year | US$ 12820 million |
| Forecasted market size in 2031 | US$ 15660 million |
| CAGR | 2.9% |
| Base Year | year |
| Forecasted years | 2025 - 2031 |
| Segment by Type |
|
| Segment by Application |
|
| Consumption by Region |
|
| By Company | Dermalogica, Environ SkinCare, Nimue, Babor, Guinot, Olay, Marykay, Artistry, Aupres, L’Oreal, Estee Lauder, Lancome, Longrich, Chcedo, Vichy, Shiseido, Fancl, Dior, SKII, Clinique, Unilever, Procter&Gamble, Biotherm, Kiehl |
| Forecast units | USD million in value |
| Report coverage | Revenue and volume forecast, company share, competitive landscape, growth factors and trends |