What is Intravenous Surgical Devices - Global Market?
Intravenous surgical devices are essential tools used in medical procedures that involve the administration of fluids, medications, or nutrients directly into a patient's vein. The global market for these devices is vast and continually evolving, driven by advancements in medical technology and an increasing demand for minimally invasive procedures. These devices include a range of products such as venous stents, catheters, and guidewires, each serving a specific purpose in the medical field. The market's growth is fueled by factors such as the rising prevalence of chronic diseases, an aging population, and the increasing number of surgical procedures worldwide. Additionally, the development of innovative products and the expansion of healthcare infrastructure in emerging economies contribute to the market's expansion. As healthcare providers strive to improve patient outcomes and reduce hospital stays, the demand for efficient and reliable intravenous surgical devices continues to rise. This market is characterized by intense competition among manufacturers, who are constantly seeking to enhance their product offerings through research and development. Overall, the global market for intravenous surgical devices plays a crucial role in modern healthcare, providing essential tools that enable medical professionals to deliver effective and timely care to patients.
Venous Stents, Catheters, Guidewires, Others in the Intravenous Surgical Devices - Global Market:
Venous stents, catheters, guidewires, and other intravenous surgical devices are integral components of the global healthcare landscape. Venous stents are small, expandable tubes inserted into veins to keep them open, ensuring proper blood flow. They are commonly used in patients with conditions such as deep vein thrombosis or chronic venous insufficiency. The demand for venous stents is increasing due to the rising incidence of vascular diseases and the growing preference for minimally invasive procedures. Catheters, on the other hand, are flexible tubes inserted into the body to deliver or remove fluids. They are used in a variety of medical settings, from administering medications to draining urine. The versatility of catheters makes them indispensable in both acute and chronic care settings. Guidewires are thin, flexible wires that help navigate through blood vessels during procedures such as angioplasty. They provide support and guidance for the placement of other devices, such as stents or catheters. The precision and reliability of guidewires are crucial for the success of many surgical interventions. Other intravenous surgical devices include infusion pumps, which control the delivery of fluids and medications, and needles, which are used for accessing veins. The global market for these devices is driven by technological advancements, increasing healthcare expenditure, and the growing prevalence of chronic diseases. Manufacturers are focusing on developing innovative products that offer improved safety, efficacy, and ease of use. Additionally, the expansion of healthcare infrastructure in emerging economies presents significant growth opportunities for the market. As the demand for minimally invasive procedures continues to rise, the market for venous stents, catheters, guidewires, and other intravenous surgical devices is expected to grow steadily. These devices play a vital role in modern medicine, enabling healthcare professionals to perform complex procedures with precision and efficiency. The competition among manufacturers is intense, with companies investing heavily in research and development to gain a competitive edge. Overall, the global market for intravenous surgical devices is poised for significant growth, driven by the increasing demand for advanced medical technologies and the need for effective and efficient patient care.
Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Others in the Intravenous Surgical Devices - Global Market:
Intravenous surgical devices are widely used across various healthcare settings, including hospitals, ambulatory surgical centers, specialty clinics, and other medical facilities. In hospitals, these devices are essential for a wide range of procedures, from routine blood tests to complex surgeries. Hospitals rely on intravenous surgical devices to ensure the safe and effective delivery of medications, fluids, and nutrients to patients. The use of these devices helps reduce the risk of complications and improves patient outcomes. In ambulatory surgical centers, intravenous surgical devices are used to perform minimally invasive procedures that do not require an overnight hospital stay. These centers offer a cost-effective alternative to traditional hospital-based surgeries, providing patients with access to high-quality care in a convenient setting. The use of intravenous surgical devices in ambulatory surgical centers is increasing as more patients seek outpatient procedures for conditions such as varicose veins or hernias. Specialty clinics, such as oncology or cardiology centers, also rely heavily on intravenous surgical devices to deliver targeted treatments to patients. These clinics often use advanced devices, such as drug-eluting stents or specialized catheters, to provide personalized care to patients with complex medical conditions. The use of intravenous surgical devices in specialty clinics is driven by the need for precise and effective treatment options that improve patient outcomes. Other medical facilities, such as nursing homes or rehabilitation centers, also use intravenous surgical devices to provide care to patients with chronic conditions or those recovering from surgery. The versatility and reliability of these devices make them indispensable in a variety of healthcare settings. Overall, the global market for intravenous surgical devices is driven by the increasing demand for advanced medical technologies and the need for efficient and effective patient care across various healthcare settings. As healthcare providers continue to seek ways to improve patient outcomes and reduce costs, the use of intravenous surgical devices is expected to grow, providing significant opportunities for manufacturers and healthcare providers alike.
Intravenous Surgical Devices - Global Market Outlook:
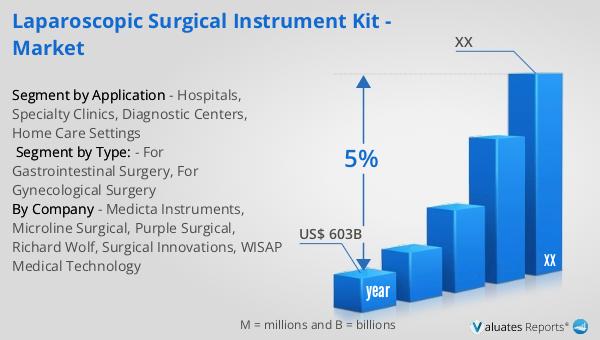
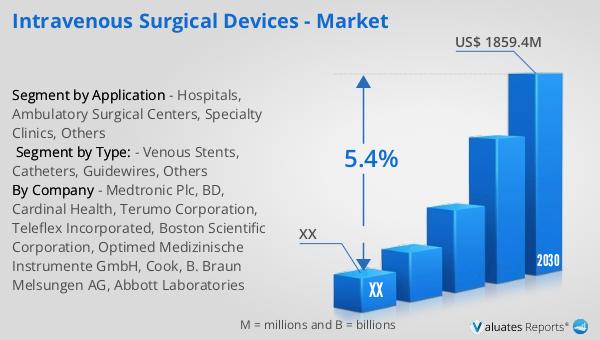
The global market for intravenous surgical devices was valued at approximately $1,249.9 million in 2023. It is projected to grow to a revised size of $1,859.4 million by 2030, reflecting a compound annual growth rate (CAGR) of 5.4% during the forecast period from 2024 to 2030. This growth is indicative of the increasing demand for advanced medical devices that facilitate efficient and effective patient care. The broader medical devices market, estimated at $603 billion in 2023, is also expected to grow at a CAGR of 5% over the next six years. This growth is driven by factors such as technological advancements, an aging population, and the rising prevalence of chronic diseases. The intravenous surgical devices market is a significant segment of the overall medical devices industry, playing a crucial role in modern healthcare. As healthcare providers continue to seek ways to improve patient outcomes and reduce costs, the demand for reliable and efficient intravenous surgical devices is expected to rise. Manufacturers are focusing on developing innovative products that offer improved safety, efficacy, and ease of use, further driving market growth. The expansion of healthcare infrastructure in emerging economies also presents significant opportunities for the market. Overall, the global market for intravenous surgical devices is poised for significant growth, driven by the increasing demand for advanced medical technologies and the need for effective and efficient patient care.
| Report Metric | Details |
| Report Name | Intravenous Surgical Devices - Market |
| Forecasted market size in 2030 | US$ 1859.4 million |
| CAGR | 5.4% |
| Forecasted years | 2024 - 2030 |
| Segment by Type: |
|
| Segment by Application |
|
| By Region |
|
| By Company | Medtronic Plc, BD, Cardinal Health, Terumo Corporation, Teleflex Incorporated, Boston Scientific Corporation, Optimed Medizinische Instrumente GmbH, Cook, B. Braun Melsungen AG, Abbott Laboratories |
| Forecast units | USD million in value |
| Report coverage | Revenue and volume forecast, company share, competitive landscape, growth factors and trends |